Greener chemistry for amide synthesis in drug development
Merging biocatalysis and organocatalysis in a single cascade
3 April 2026
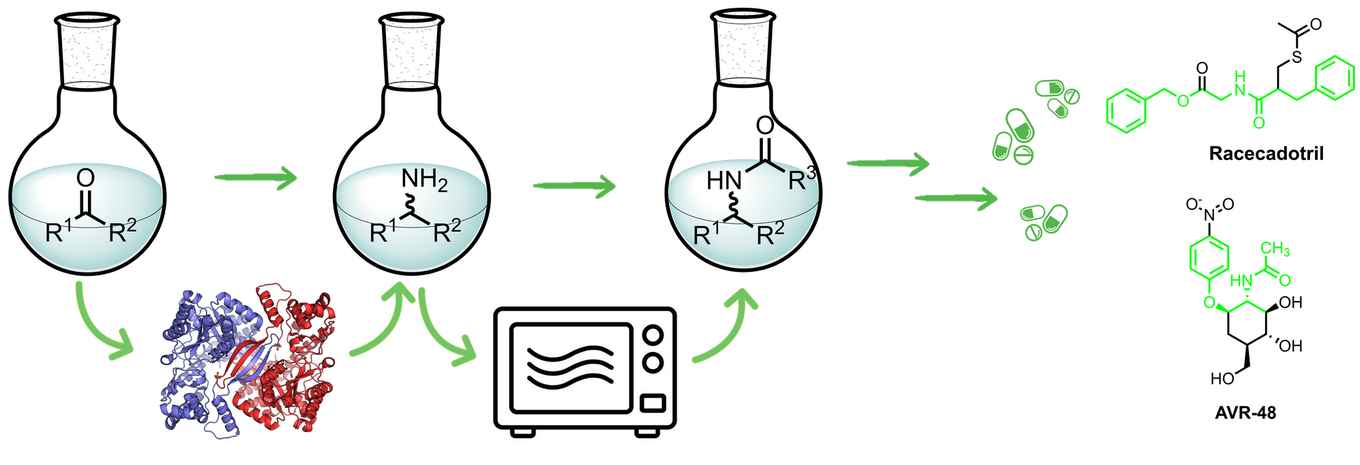
The method presented in ChemSusChem successfully integrates biocatalysis and organocatalysis into a single cascade process for the synthesis of chiral amides. The methodology offers a greener alternative to conventional amidation strategies, which often rely on toxic reagents and generate significant waste. Its applicability to pharmaceutical synthesis is highlighted by the preparation of key building blocks for drugs such as Racecadotril and AVR-48.
The presented bio-organocatalytic cascade combines ω-transaminase-catalyzed asymmetric reductive amination with a choline chloride-mediated direct amidation. Starting from prochiral carbonyl compounds, the ω-transaminases enable the formation of enantiomerically pure amines (ee > 99%). These are then converted into amides using carboxylic acids in choline, the latter of which acts as both catalyst and reaction medium under microwave irradiation. Notably, removal of the acetone by-product after the first step led to a substantial improvement in amide yields, reaching 60–86%. The work demonstrates how the combination of biocatalysis with choline chloride-mediated organocatalytic amidation can be effectively leveraged to access chiral amide intermediates relevant to active pharmaceutical ingredients (APIs).
Combining complementary expertise for greener chemistry
The research was carried out at the HIMS-Biocat group at the University of Amsterdam, in collaboration with the Università “Magna Graecia” di Catanzaro and with additional contributions from the Vrije Universiteit Amsterdam. This collaboration brought together complementary expertise in enzymatic catalysis, organocatalysis, and process development. Although no industrial partners were directly involved, the methodology was developed with potential industrial applications in mind, particularly for the sustainable synthesis of API intermediates.
The research was primarily supported by the Marie Skłodowska-Curie Actions, with additional contributions from the Dutch Research Council (NWO) and the Fonds de la Recherche Scientifique (FNRS), underscoring its relevance within the European research landscape. The Marie Skłodowska-Curie Actions (MSCA) Postdoctoral Fellowship is a European funding program that supports researchers in gaining advanced training, international mobility, and career development through high-quality research projects in academia or industry.
Paper details
Salvatore Romano, Matteo Damian, Monica Nardi, Antonio Procopio, Sebastian Strähler, Daniël Preschel, Manuela Oliverio, Francesco G. Mutti: Stereoselective Bio-Organocatalytic Cascade to Chiral Amides as Active Pharmaceutical Ingredient Intermediates Using ω-Transaminase and Choline Chloride Under Microwave Irradiation. ChemSusChem, 2026, e202501501. DOI: 10.1002/cssc.202501501