Cofactor switches catalyst
16 January 2019
Nature’s chemistry is very efficient and ingenious. In a cell, containing a complex mixture of chemicals, enzymes ensure that chemical transformations occur at the right place and at the right moment. To control this natural catalysis, small signaling molecules and cofactors are present that regulate the function of the enzymes.
In synthetic chemical processes, reactions in complex mixtures are becoming increasingly important, for instance in the production of pharmaceutical compounds. One of the reasons for this is the transition to renewable feedstock originating from biomass. Whereas petrochemical feedstock (derived from oil) has a reasonably homogeneous composition, renewable feedstock from biomass contains a much larger variety of molecules and functionalities. For that reason it is crucial to have full control over the timing of the catalyst's activity, so that unwanted side reactions and the production of undesired products can be prevented. In homogeneous catalysis, where the catalyst is dissolved – just as enzymes are, turning on a catalyst in the middle of a process is easier said than done.
Capsule
In the ‘Homogeneous, Supramolecular and Bio-inspired Catalysis’ group of professor Joost Reek one of the research programs is focused on looking at nature as an inspiration for the design of homogeneous catalysts. In this context, PhD-student Lukas Jongkind has developed a catalyst which can be activated by the addition of a small ‘cofactor’, forming a highly selective catalyst after activation. This work was carried out in collaboration with the Radboud Universiteit Nijmegen.
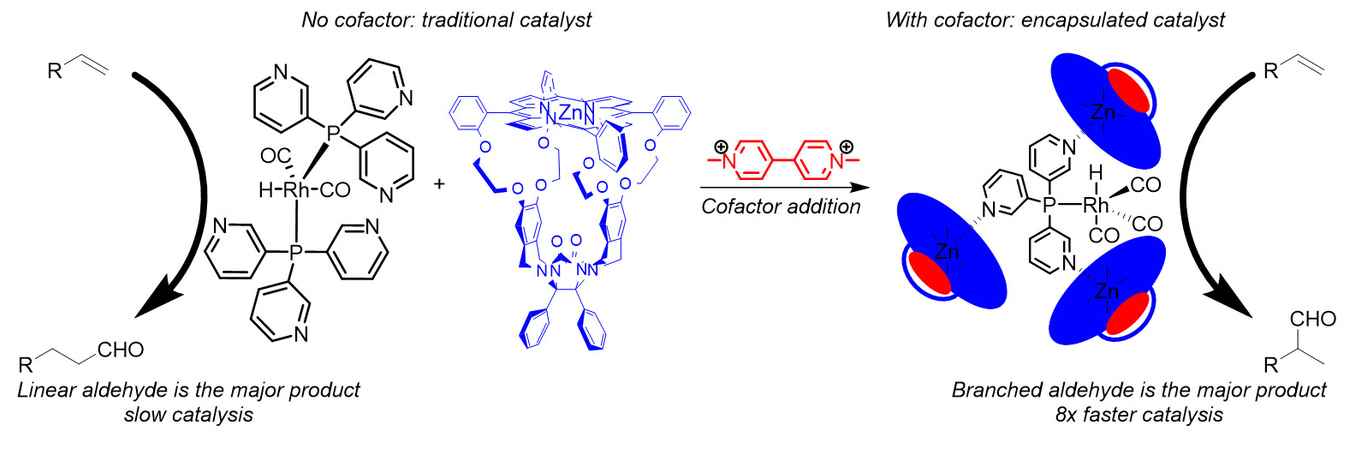
The catalyst consists of supramolecular building blocks for which the interactions can be regulated by means of a cofactor. Without the cofactor there is negligible interaction, resulting in an inefficient catalyst. When the cofactor is added, seven building blocks self-assemble, forming a capsule around a catalytically active rhodium atom present in solution. The capsule resembles both in shape and function the structure around the active site of enzymes, enabling selective chemical processes.
Lukas Jongkind investigated the application of the catalyst in rhodium catalyzed hydroformylation, a process which converts alkenes to aldehydes. Hydroformylation is one of the most abundant chemical processes in which a homogeneous catalyst is applied. In this reaction the regulation of the catalyst by addition of the cofactor proves to be very successful: after addition of the cofactor the formation of the capsule around the rhodium atom increases the activity of the catalysts eightfold. In addition, a different aldehyde is formed compared to when the cofactor is absent. The capsule therefore improves both the activity and the selectivity of the hydroformylation reaction.
New catalysts
With these results, the researchers have shown that switching of a relatively simple homogeneous catalysts is possible through a process resembling regulation in enzymes. This provides opportunities for the development of new homogeneous catalysts that can operate selectively in complex chemical mixtures.
Publication details
Lukas J. Jongkind, Johannes A.A.W. Elemans, Joost Reek: Cofactor controlled encapsulation of a rhodium hydroformylation catalyst. Angewandte Chemie, First published: 09 January 2019. DOI: 10.1002/anie.201812610