Novel advanced MOF/polymer composite allows for tuneable separation and low cost regeneration
21 October 2019
To meet the demand for green chemistry and sustainable development, industry critically depends on new innovative processes that use materials and energy more efficiently. Much room for improvement lies in the field of separation, that accounts for 40-90% of capital and operating costs in industry. It mainly involves traditional separation technologies, such as distillation and crystallization. These account for more than 90% of all the separation processes in the chemical industry and are expensive and energy-inefficient.
Tuning novel adsorbents for efficient separation
Adsorptive separation methods can provide a cost-effective alternative here. Their separation efficiency relies on the internal porosity and surface properties of solid adsorbent materials. Compared to traditional adsorbents, such as zeolites and carbon-based materials, metal-organic frameworks (MOFs) are a very promising class of novel customisable porous materials.
Now, HIMS PhD student Yiwen Tang, under the supervision of Dr Stefania Grecea and Dr David Dubbeldam, has introduced a new concept in tailoring MOFs. He has shown that introducing organic polymers within the pores of metal-organic frameworks enables to fine-tune the pore size and thus enhance the separation of water-alcohol mixtures.
In addition, by carefully choosing the type of polymer, Tang was able to control the desorption of water and alcohol molecules, therefore enabling an efficient and cost-effective regeneration process. To achieve this, he made use of the phenomenon that a certain type of organic polymers can undergo a phase transition between the hydrophobic and hydrophilic phases in response to a temperature change.
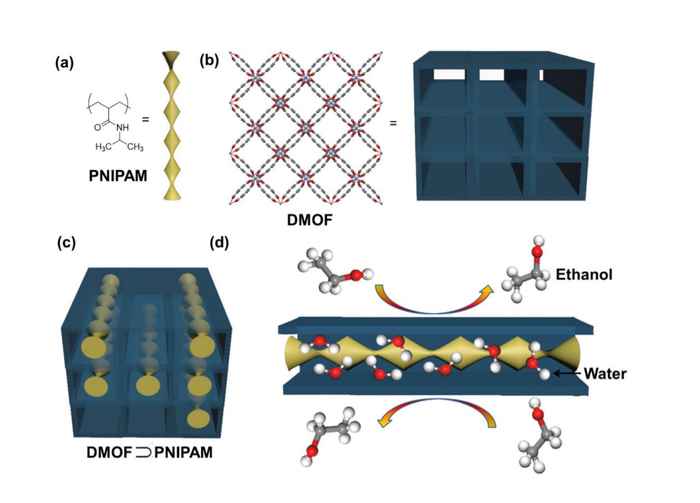
Loading a MOF with amide polymers
In a recent paper in the ACS publication Applied Materials & Interfaces, the researchers show that introducing polymer chains containing amide groups within the pores of a hydrophobic metal-organic framework not only allows to control the pore’s size of the adsorbent material, but also to tune the water uptake. The latter can be enhanced by increasing the loading of the MOF with the polymer, which implies that the exposed amide groups of the polymer gradually alter the hydrophobicity of the composite.
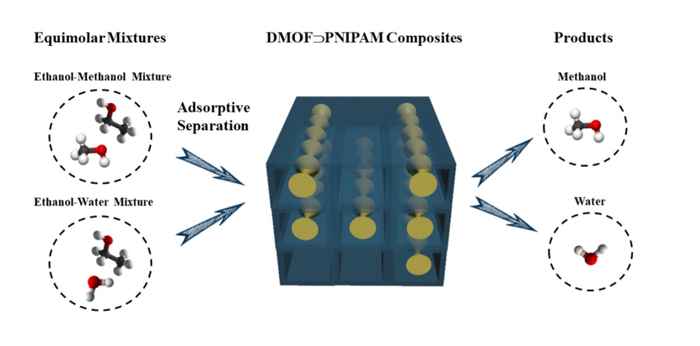
The researchers established that the MOF/polymer composite with the highest polymer loading displays selective adsorption for water and methanol over ethanol when using equimolar mixtures of methanol-ethanol and water-ethanol. They confirmed this by single-component adsorption measurements as well as molecular simulations. Notably, the water stability of the metal-organic framework was greatly improved after the incorporation of the organic polymer in its pores.
Moreover, it was demonstrated that the composite can be regenerated at 60 °C and can be reused in consecutive water-methanol separation cycles. This is a tremendous improvement in the regeneration of adsorbent materials. In particular in comparison to zeolites, which require a thermal treatment at around 200−300 °C for the desorption of water molecules, it will lead to a significant increase of the energy efficiency of the overall separation process.
The research was supported by the Research Priority Area Sustainable Chemistry, and the Chinese Scientific Council (through a CSC PhD fellowship to Yiwen Tang).
Publication details
Yiwen Tang, David Dubbeldam, Stefania Tanase: Water-ethanol and methanol-ethanol separations using in situ confined polymer chains in a metal-organic framework. ACS Appl. Mater. Interfaces. Published online: 10 October 2019. DOI: 10.1021/acsami.9b1436
Researchers' webpages:
Read also:
Looking at leaves: enhancing the performance of MOF materials.